Having seen the impact of cell-free DNA technology across various fields, I recognize its powerful potential in transforming patient care.



Having seen the impact of cell-free DNA technology across various fields, I recognize its powerful potential in transforming patient care.
David Wojciechowski
Professor of Medicine,
Medical Director Kidney Transplantation
Dallas, TX
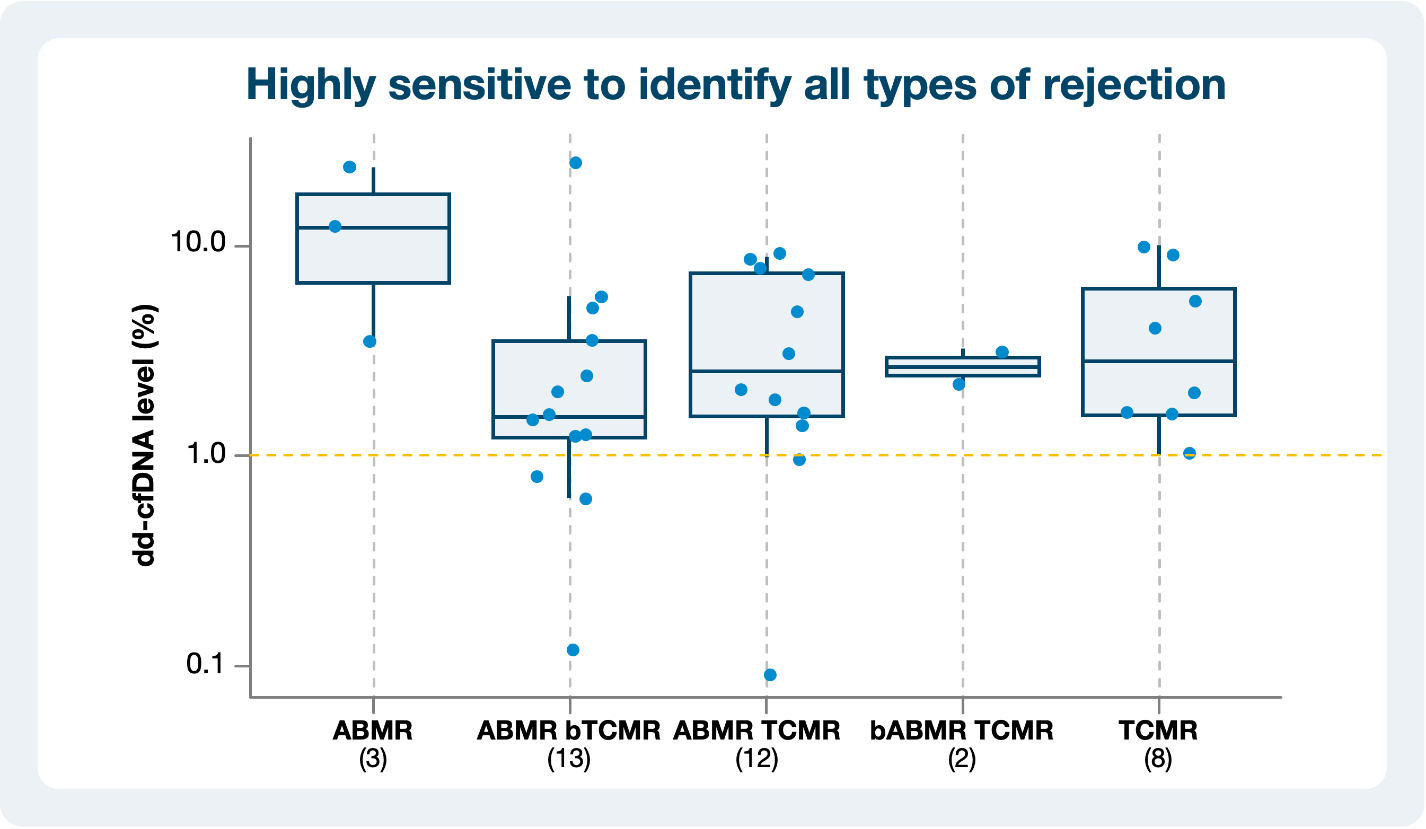
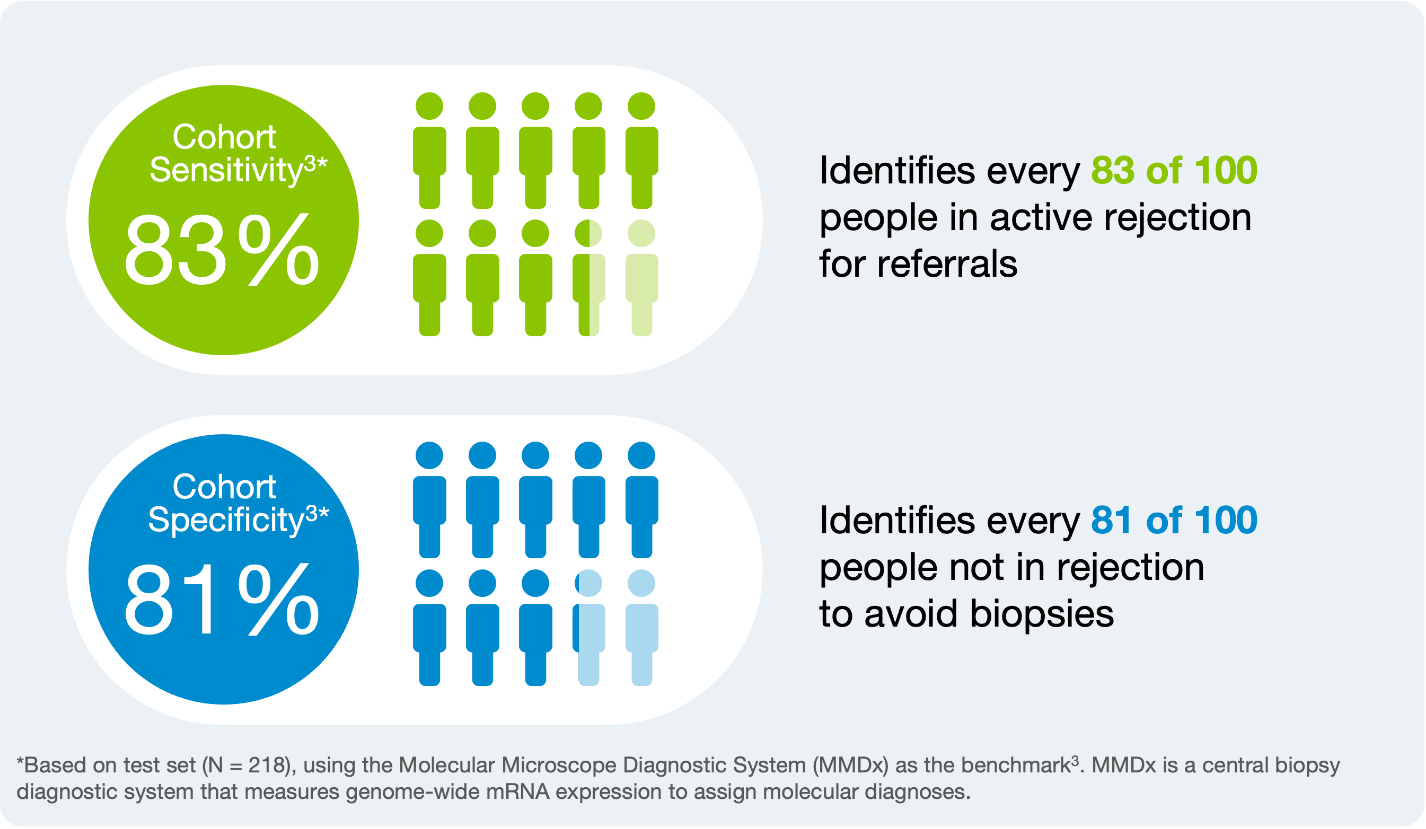
ProsperaTM is a proven leading indicator of rejection1-3
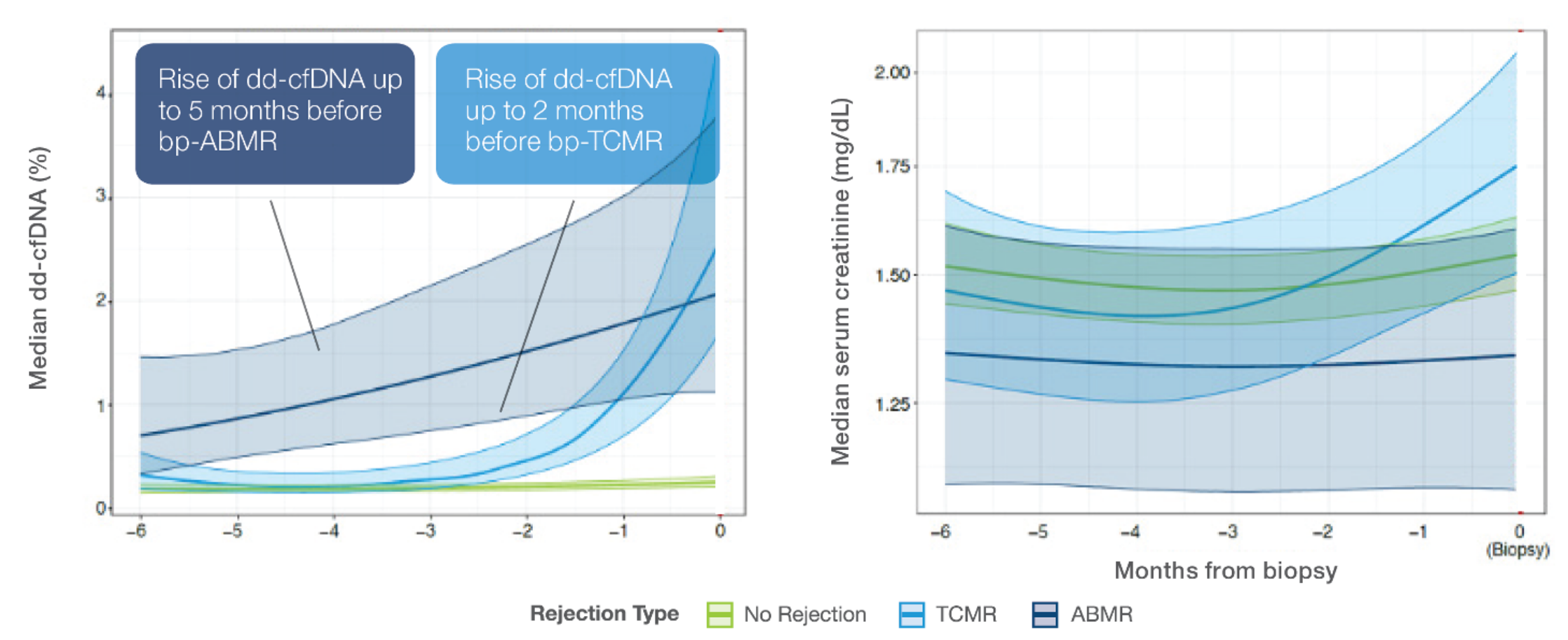
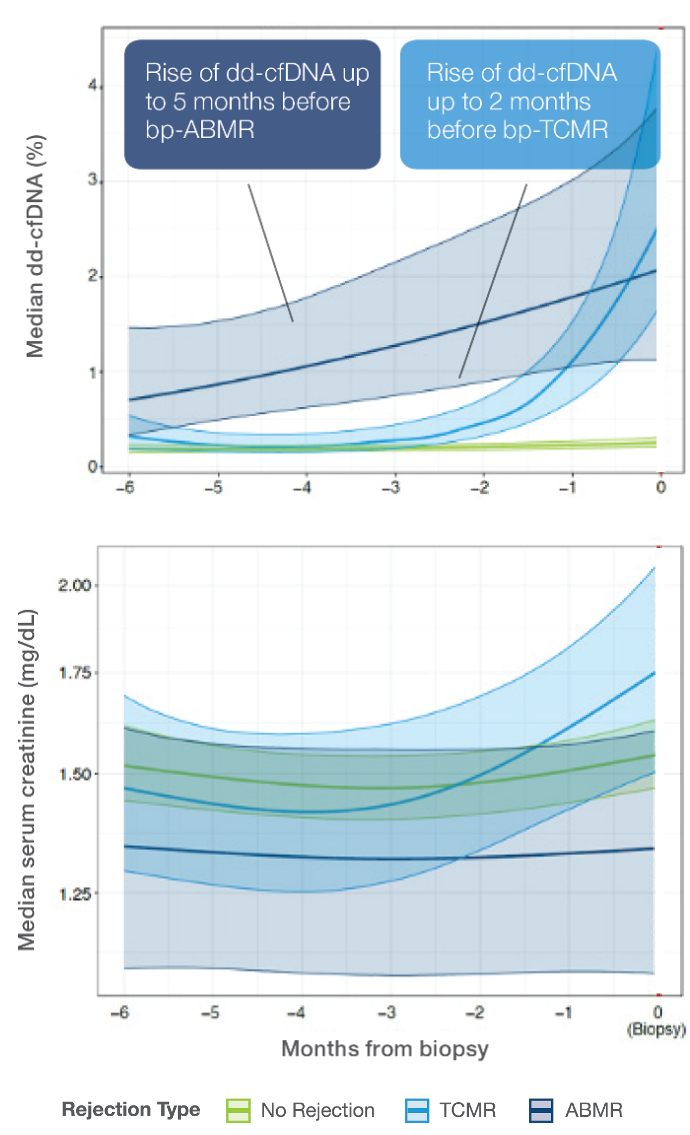
Predicted antibody-mediated rejection (ABMR) up to five months and T cell-mediated rejection (TCMR) up to two months in advance of biopsy-proven rejection1
Increased ProsperaTM levels in non-rejecting patients are associated with worsening clinical graft function
Consistent ProsperaTM performance across validations and real-world settings to benefit any patient population1-3
The ProsperaTM test predicted ABMR up to 5 months and TCMR up to 2 months in advance of biopsy-proven rejection1
ProsperaTM dd-cfDNA levels versus serum creatinine prior to biopsy-proven rejection
Increased ProsperaTM levels in non-rejecting patients are associated with worsening clinical graft function1,4

Two or more elevated ProsperaTM tests in a patient were correlated with worse eGFR.
This observation is consistent with recent findings indicating that elevated ProsperaTM result in non-rejection biopsies is often associated with sub-threshold ABMR and should therefore not be considered a false positive.
The interim ProActive study data solidifies ProsperaTM use as a non-invasive surveillance tool1,2,5,6
~9 out of every 10 clinically stable patients had consistently low ProsperaTM dd-cfDNA levels <1%5
Elevation of cfDNA from baseline when using serial ProsperaTM testing indicated increased risk of rejection6
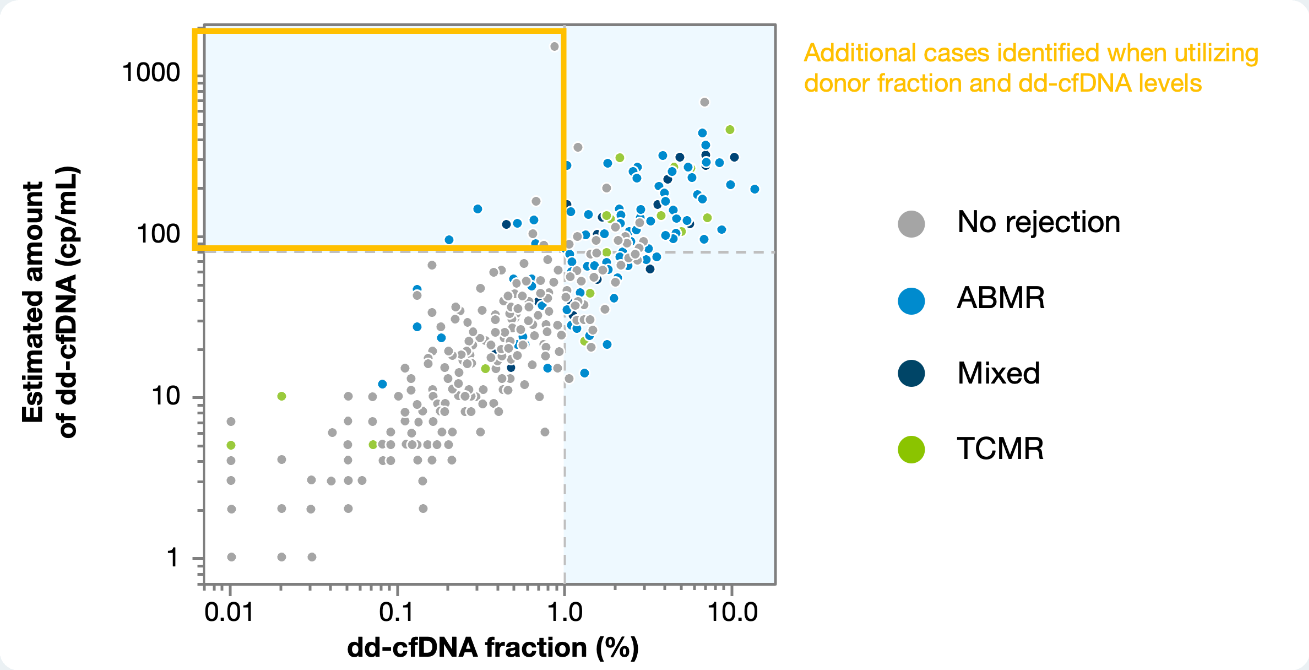
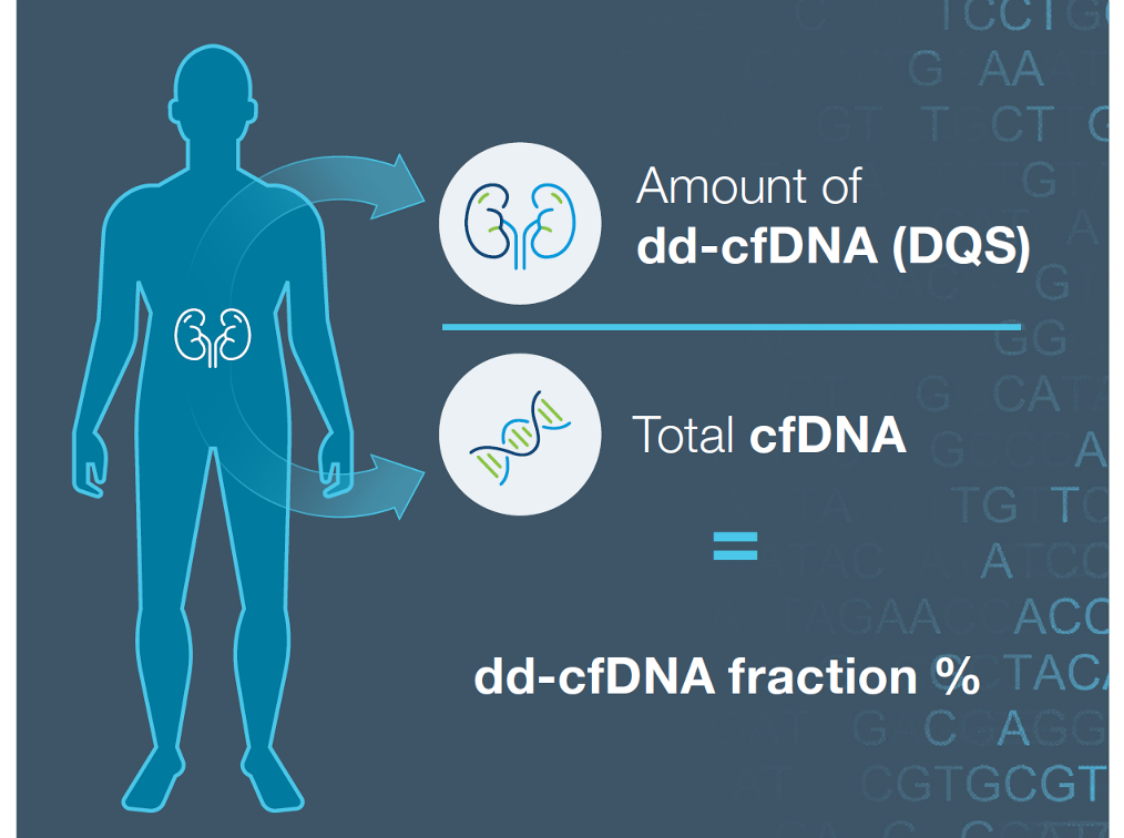
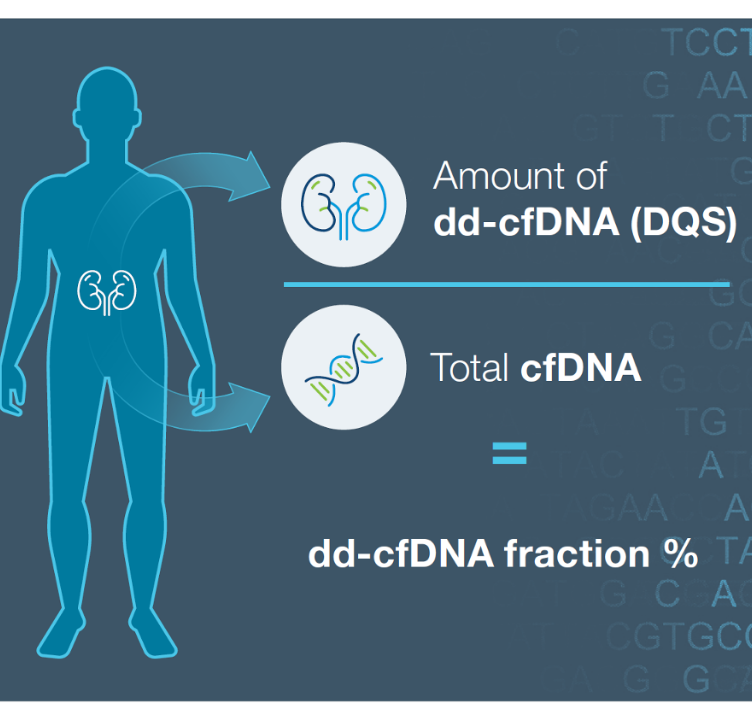
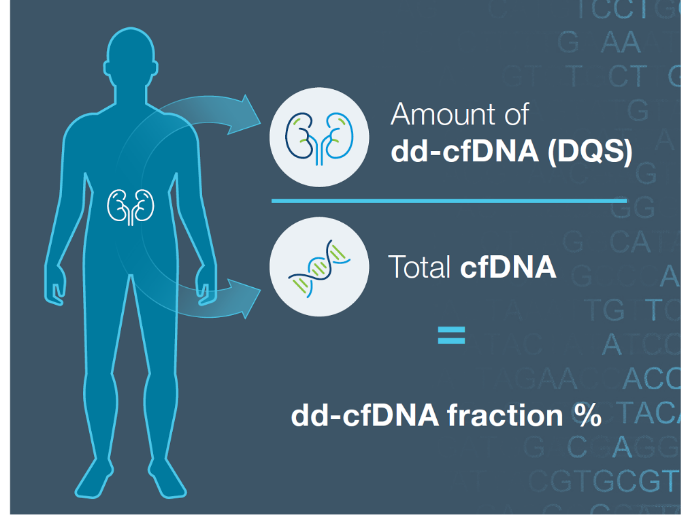
ProsperaTM surveillance testing in action: The Donor Quantity Score (DQS) difference in patient care
Case: Diagnosing ABMR due to serial ProsperaTM with DQS finding
Patient
demographics
- Female in mid-70s
- High risk due to high CPRA; previous history of biopsy-proven ABMR
ProsperaTM test
summary
| ProsperaTM Draw | ~1 year post-tx |
~13 months post-tx |
~16 months post-tx |
~18 months post-tx |
~18.5 months post-tx |
|---|---|---|---|---|---|
| Donor Fraction Estimate (DFE) | 0.1% | 0.1% | 0.32% | 0.3% | 0.6% |
| Donor Quantity Score (DQS) | 7.9 cp/mL | 12.4 cp/mL | 15.9 cp/mL | 88.0 cp/mL | 83.5 cp/mL |
| Biopsy diagnosis | ABMR |
| ProsperaTM Draw | Donor Fraction Estimate (DFE) | Donor Quantity Score (DQS) | Biopsy diagnosis |
| ~1 year post-tx | 0.1% | 7.9 cp/mL | |
| ~13 months post-tx | 0.1% | 12.4 cp/mL | |
| ~16 months post-tx | 0.32% | 15.9 cp/mL | |
| ~18 months post-tx | 0.3% | 88.0 cp/mL | |
| ~18.5 months post-tx | 0.6% | 83.5 cp/mL | ABMR |
Key Takeaway
Repeat testing with ProsperaTM revealed consecutive results above DQS threshold, prompting a biopsy to confirm rejection. Test with only donor fraction results may not have triggered the necessary follow-up to find this case of ABMR.
Case: Diagnosing TCMR ~9 months post-transplant due to ProsperaTM with DQS finding
Patient
demographics
- Female in mid-50s
- High risk due to COVID-19, BK infection, DSA+, and allograft dysfunction
ProsperaTM test
summary
| ProsperaTM Draw | ~2 months post-tx |
~3 months post-tx |
~4 months post-tx |
~6 months post-tx |
~9 months post-tx |
|---|---|---|---|---|---|
| Donor Fraction Estimate (DFE) | 0.1% | 0.1% | 0.07% | 0.08% | 0.43% |
| Donor Quantity Score (DQS) | 6.0 | 11.3 | 8.7 | 6.0 | 92.6 |
| Biopsy diagnosis | TCMR |
| ProsperaTM Draw | Donor Fraction Estimate (DFE) | Donor Quantity Score (DQS) | Biopsy diagnosis |
| ~2 months post-tx | 0.1% | 6.0 | |
| ~3 months post-tx | 0.1% | 11.3 | |
| ~4 months post-tx | 0.07% | 8.7 | |
| ~6 months post-tx | 0.08% | 6.0 | |
| ~9 months post-tx | 0.43% | 92.6 | TCMR |
Key Takeaway
ProsperaTM with DQS flagged an otherwise undetected issue that led to diagnosis of TCMR. The donor fraction estimate was elevated, yet was still far below the threshold for rejection and likely would not have led to follow-up as quickly.
Case: Diagnosing TCMR at first year post-transplant due to ProsperaTM with DQS finding
Patient
demographics
- Male in mid-50s
- Long time on dialysis pre-transplant and type II diabetes
ProsperaTM test
summary
| ProsperaTM Draw | ~1.5 months post-tx |
~3 months post-tx |
~3.5 months post-tx |
~6 months post-tx |
~1 year post-tx |
|---|---|---|---|---|---|
| Donor Fraction Estimate (DFE) | 0.12% | 0.14% | 0.1% | 0.1% | 0.48% |
| Donor Quantity Score (DQS) | 9.5 | 8.5 | 6.2 | 5.0 | 95.0 |
| Biopsy diagnosis | TCMR |
| ProsperaTM Draw | Donor Fraction Estimate (DFE) | Donor Quantity Score (DQS) | Biopsy diagnosis |
| ~1.5 months post-tx | 0.12% | 9.5 | |
| ~3 months post-tx | 0.14% | 8.5 | |
| ~3.5 months post-tx | 0.1% | 6.2 | |
| ~6 months post-tx | 0.1% | 5.0 | |
| ~1 year post-tx | 0.48% | 95.0 | TCMR |
Key Takeaway
Even with a stable serial donor fraction estimate, a significant DQS increase in the ProsperaTM result prompted diagnosis of TCMR.
De-identified real patient cases for illustrative purposes.
bp-ABMR=biopsy-proven antibody-mediated rejection; bp-TCMR= borderline T cell-mediated rejection; eGFR=epidermal growth factor receptor.
References:
- Bromberg JS, Bunnapradist S, Samaniego-Picota M, et al. Elevation of donor-derived cell-free DNA before biopsy-proven rejection in kidney transplant. Transplantation. 2024;108(9):1994-2004. doi:10.1097/TP.0000000000005007
- Sigdel TK, Archila FA, Constantin T, et al. Optimizing detection of kidney transplant injury by assessment of donor-derived cell-free DNA via massively multiplex PCR. J Clin Med. 2018;8(1):19. doi:103390/jcm8010019
- Halloran PF, Reeve J, Madill-Thomsen KS, et al. Combining donor-derived cell-free DNA fraction and quantity to detect kidney transplant rejection using molecular diagnoses and histology as confirmation. Transplantation. 2022;106(12):2435-2442. doi:10.1097/TP.0000000000004212
- Gauthier PT, Madill-Thomsen KS, Demko Z, et al. Distinct molecular processes mediate donor-derived cell-free DNA release from kidney transplants in different disease states. Transplantation. 2024;108(4):898-910. doi:10.1097/TP.0000000000004877
- Bunnapradist S, et al. Increases from baseline dd-cfDNA fraction are associated with acute allograft rejection in kidney transplant recipients. Poster presented at: American Transplant Congress; June 3-7, 2023; San Diego, CA.
- Cooper M, Chen Y-A, Barnes D, et al. Donor-derived cfDNA fraction levels in stable kidney transplant recipients in the first year after transplant. Poster presented at: American Transplant Congress; June 3-7, 2023; San Diego, CA.